Defining LEL (Lower Explosive Limit) as it Pertains to Calibration Gas
If you spend much time working with calibration gas, it’s very likely that you will see the term “LEL” used extensively. LEL, short for “Lower Explosive Limit“, is defined as the lowest concentration (by percentage) of a gas or vapor in air that is capable of producing a flash of fire in presence of an ignition source (arc, flame, heat, etc.). Concentrations lower than the Lower Explosive Limit are ‘too lean’ to burn; those above the Upper Explosive Limit (UEL) are too rich to burn.
In gas-detection systems, the amount of gas present is specified as a percentage (%) of LEL. Zero percent Lower Explosive Limit (0% LEL) denotes a combustible gas-free atmosphere. One hundred percent lower explosive limit (100% LEL) denotes an atmosphere in which gas is at its lower flammable limit. The relationship between percent LEL and percent by volume differs from gas to gas. The example below demonstrates the flammability of Methane (Natural Gas) in Air. In concentrations of 0-5% Methane in air, the mixture is too lean to ignite or burn. Methane concentrations between 5% and 17% in will support ignition and are considered highly flammable. At levels above 17%, the atmosphere is too rich for the methane to ignite.
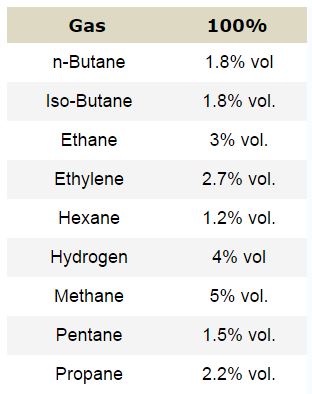
To compute the LEL of any gas in air, divide the unknown concentration by the LEL listed in the NFPA Handbook. 100% LEL’s for 9 of Gasco’s more common gasses are shown in the Table 1A here. For example, if you take 2.5% Methane in air and divide it by 100% LEL of methane (5%), the result is 50% LEL. (2.5% ÷ 5% = 50%) The same theory can be applied to any of the gasses listed here. Conversely, you can multiply the % LEL of the unknown concentration by the 100% LEL to obtain the % by volume. (50% LEL methane x 5% = 2.5% volume).
It’s also important to remember that in order to ignite and burn, a fire requires three elements: heat, fuel, and an oxidizing agent (usually oxygen). The fire can be prevented or extinguished by removing any one of these elements.
Since air is made up primarily of 20.9% Oxygen with the balance being nitrogen, if you take Oxygen out of the equation, the methane is no longer flammable at any concentration. This is important when ordering mixtures with no oxygen content; without oxygen, LEL is not a factor.
If you need help determining the correct calibration gas for your application, our experts are here to help!
