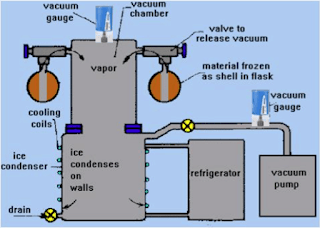
Lyophilization, also known as cryodesiccation, or more commonly “freeze-drying” is a dehydration process typically used to preserve a perishable material or make the material more convenient for transport. The freeze-drying process works by exposing a material that has been frozen to a vacuum environment. This lowers the vapor pressure of the frozen water contained in the material. Heat will be applied to the environment, driving the water out of the material through the process of sublimation.
In order to avoid the liquid phase of the frozen water, it is absolutely essential to lower the partial pressure of water, below the triple point pressure. The triple point is the point where 3 phases – liquid, solid, and vapor – coexist at a particular temperature and pressure. As the water vapor exits the sample, a chilled surface known as the ice condenser collects the vapor from the evolving product capturing it and preventing reabsorption or saturation of the chamber environment.
Applications
BioPharm manufacturing: Uses lyophilization to preserve products such as vaccines and other injectable drugs (parenterals). The removal of water from these materials allows for the preservation, shipping, and storage of smaller volumes of material. Tablets or wafers are also freeze dried often creating dosage forms which are more rapidly absorbed or more easily administered. Proteins, enzymes, microorganisms, and blood plasma are also examples of materials that are commonly lyophilized. In bioseparation processes, lyophilization is also used as a method to remove solvents that may be too costly or too difficult to remove by other means. FDA requires a higher temperature process manometer to be used in conjunction with a pirani gauge to understand when the “drying” is complete.
Food manufacturing: “Freeze-dried” foods help food manufacturers increase product shelf life and allows them to cater to the needs of consumers who are interested in convenience. The foods are typically easier to store and transport due to the removal of water and its associated weight. Foods last longer and can be used in environments outside the norm (e.g. wilderness, military situations, etc.) Common examples are coffee, fruits, and cereals.
Process Details
Once a material has been frozen, the environment in which the material lies is placed under partial vacuum, usually a few millibars. Heat is introduced forcing the frozen water to sublimate thereby exiting the material. That water vapor is often captured on a condensing coil and preventing it from re-entering the freeze dried material. It is important to note that the use of a heated capacitance manometer will prevent the unit from acting as a condenser of the water vapor.
In the final step of the process, the vacuum in the chamber is relieved by introducing an inert gas such as nitrogen. This gas regulation may be an area where Brooks Instrument variable area flow meters (rotameters) or MFCs could be supplied. “Downstream” packaging of freeze dried materials often involves “vacuum packaging” which attempts the complete removal of oxygen from the final packaged material. This is important because oxygen is the primary factor in product spoilage.
Contact the experts at Cross Company to learn more about how we can help you control your processes. Specifically we can help you in determining if a XacTorr series Capacitance Manometer is right for you freeze-drying application.
