Brooks Instrument Resource

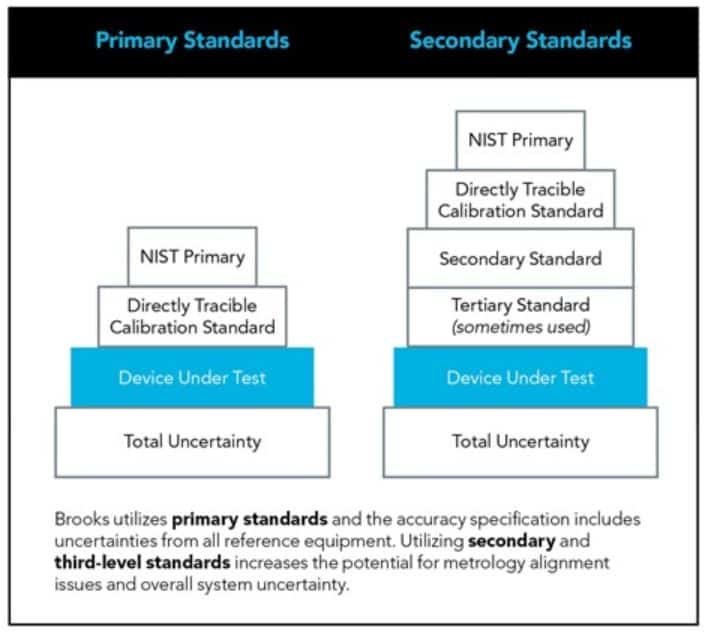
Calibration labs utilize a number of technologies and appropriate calibration reference standards to ensure your equipment is working properly. However, a basic understanding how metrology, reference standards, and calibration affect long-term performance of mass flow controllers (MFC) can help you better manage your equipment and make informed decisions.
Click below to read this article from Brooks Instrument to leran more about the basics of mass flow controller calibration.